|
We created a timeline of the history of the periodic table. The Periodic Table has constantly been improved and developed over the past 200 years, but in 1869 Dimitri Mendeleev finished the first version of the periodic table as we know it today, by arranging the elements by atomic mass and leaving spaces open for the elements that were not yet discovered. Now, let me recall to you the atomic mass of lithium, sodium and potassium. Interactive periodic table with up-to-date element property data collected from authoritative sources. The main thing I want to tell you is that he arranged these elements in the increasing order of their ATOMIC MASS. 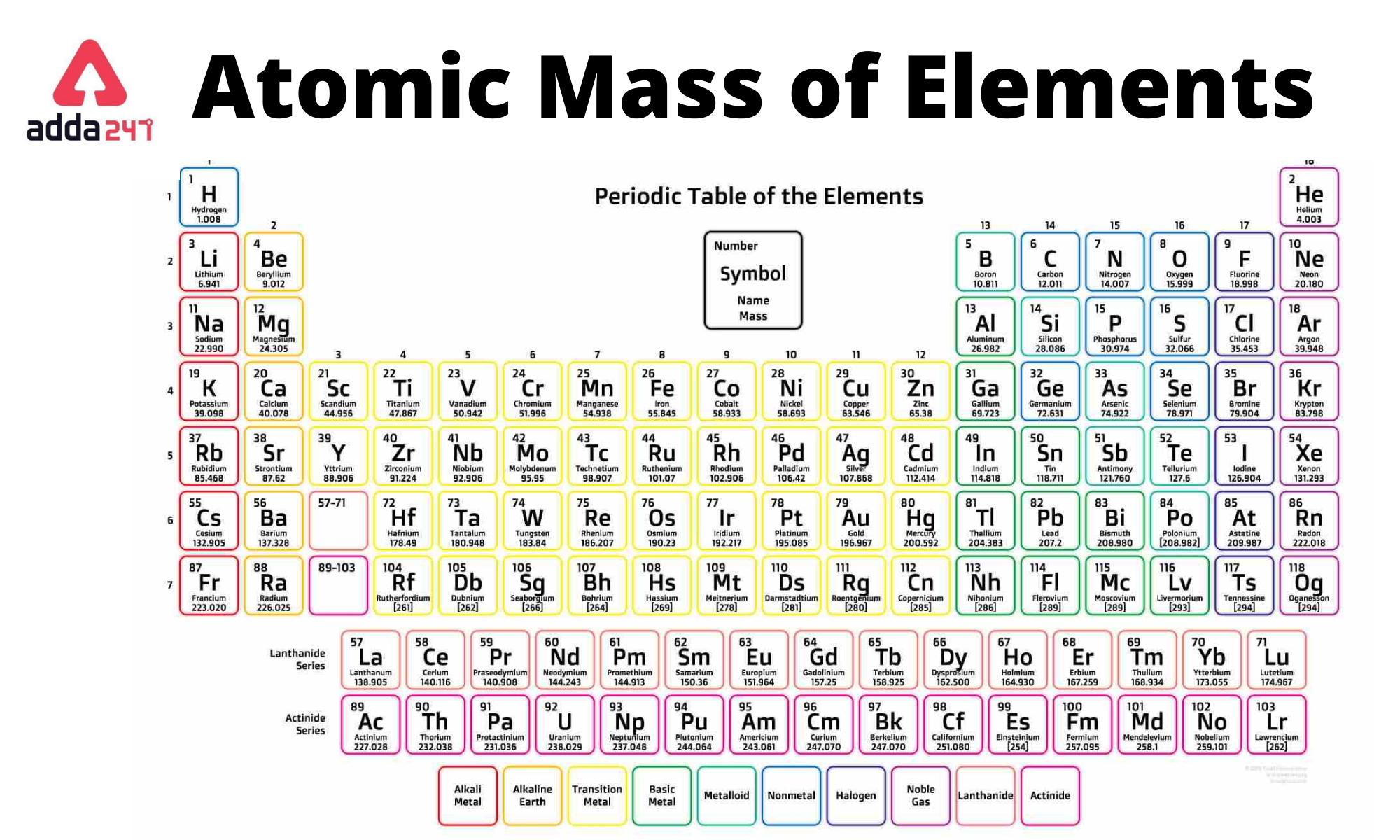
This list contains the 118 elements of chemistry. Calcium (Ca), strontium (Sr) and Barium (Ba) in other triad and, Chlorine (Cl), bromine (Br) and iodine (I) in other triad. click on any elements name for further information on chemical properties, environmental data or health effects. Who invented the Periodic Table of Elements? Periodic table Elements molar mass Chemical elements listed by atomic mass The elements of the periodic table sorted by atomic mass. The physical properties of halogens vary significantly as they can exist as solids, liquids, and gases at room temperature. Nichrome is an alloy of nickel and chromium with small amounts of silicon, manganese and iron. It is, however, mainly used in making alloys such as stainless steel. Nickel resists corrosion and is used to plate other metals to protect them. :max_bytes(150000):strip_icc()/PeriodicTablecolor-58b5d9105f9b586046df7d8a.jpg)
The term 'halogen' means 'salt-former' and compounds that contain one of the halogens are salts. A silvery metal that resists corrosion even at high temperatures. The periodic table also gives us an idea of what the characteristics of an element might be and help us predict how an element might react based on in which group it is located. They are located on group 17 of the periodic table and have a charge of -1. The Periodic Table of Elements can be used as an assisting tool in chemical calculations, when a specification of an element is needed it is easily found in the Periodic Table. The total number of atoms in a substance can also be determined by using the relationship between grams, moles, and atoms. How is the Periodic Table of Elements used? This periodic table is a periodic table with charges PDF optimized to fit on a single sheet of paper. Multiply moles of Ca by the conversion factor (molar mass of calcium) 40.08 g Ca/ 1 mol Ca, which then allows the cancelation of moles, leaving grams of Ca. 
The periodic table lists the atomic mass of carbon as 12.011 amu the average molar mass of carbonthe mass of 6. The table lists all the elements that are currently known (118), in descending order of the number of protons that are present, in a single atom of the element. For elements with no stable isotopes, the mass number of the isotope with the longest half-life is in parentheses. The molar mass of any substance is its atomic mass, molecular mass, or formula mass in grams per mole. The Periodic table of elements is a tool, developed by scientists over hundreds of years. List of all the elements and their properties:
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
